Bond Enthalpies
Image
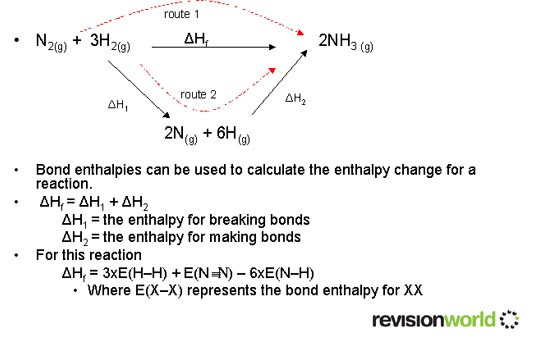
Calculation of Bond Enthalpies
Bond enthalpies are a measure of the energy required to break a bond.
They are always negative.
The values quoted are usually average values, as the strength of a bond between two atoms is dependant on the molecule.
They are difficult to measure directly they are usually calculated from data enthalpy changes of combustion of several compounds.
