What is crude oil?
Crude oil is a mixture of a very large number of compounds.
It is formed from the remains of plants and animals which died millions of years ago.
This is why it is called a fossil fuel.
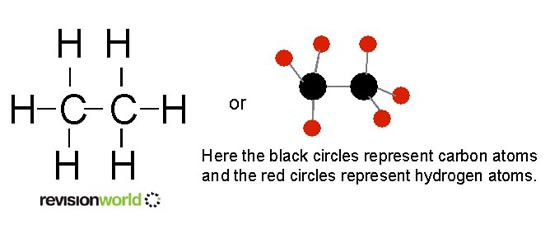
Most of the compounds in crude oil consist of molecules made up of hydrogen and carbon atoms only, we call these type of compounds hydrocarbons.
We represent hydrocarbons in the following ways:
Image
Useful products from Oil
Most fuels contain carbon and/or hydrogen and may also contain some sulphur.
The gases released into the atmosphere when a fuel burns may include:
- carbon dioxide
- water (vapour), which is an oxide of hydrogen
- sulphur dioxide (This gas dissolves in rain and forms acid rain)
This GCSE Chemistry video exaplins how fuels are made from Crude Oils
